- +91-11-9810681132, 9868532156
- info@mittalenterprises.com
Note: No part of this website may be reproduced, stored in a retrieval, or transmitted in any form or by any mean
Note: No part of this website may be reproduced, stored in a retrieval, or transmitted in any form or by any mean

Objective:
1.Characterization of Nanofluids like Ag/Au & Ferrofluids etc.
2. To evaluate modest nanoparticles concentration in the fluid for significant enhancement of its property.
3. Prediction of enhanced thermal conductivity due to suspension of the metallic nanoparticles with very low concentration in to the polymeric fluids.
4. Study of Phase transition and to detect/assess weak and strong molecular interactions in Nanofluids.

|
|||
|
Nano Fluid Nano Fluid Interferometer Request for Brochure Working Principle In this instrument, Ultrasound waves of known frequency are produced by a Piezo-Electric transducer and its wavelength is measured with digital micrometer with high accuracy within ±0.001mm. From the knowledge of frequency (f) and wave length(λ); the compressibility of nanofluid is determined by the following formula : Sound Velocity in nanofluid Where ρ is density of nanofluid. Thermal conductivity is determined by the following formula:-
where N (Avogadro's number) = 6.02×1023 and
Description
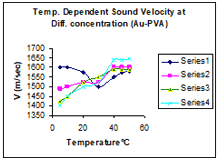
various concentrations. Models Model NF-10(2MHz ) |
|||
| Enquiry | |||